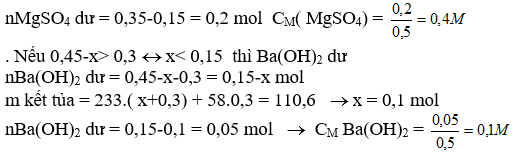DẠNG BÀI TẬP CHIA NHIỀU TRƯỜNG HỢP
- Thứ sáu - 16/10/2020 23:36
- In ra
- Đóng cửa sổ này
Bài 1: Cho rất từ từ dd A chứa x mol HCl vào dd chứa y mol Na2CO3

Sau khi cho hết A vào B được dd C. Hỏi trong C có những chất gì? bao nhiêu mol?
Giải: Khi cho từ từ HCl vào Na2CO3 xẩy ra các pư sau:
HCl + Na2CO3 ® NaHCO3 + NaCl (1)
x y y
HCl + NaHCO3 ® NaCl + CO2 + H2O (2)
x-y y
- Khi x<y : chỉ xẩy ra pư(1)
Dd C gồm x mol NaHCO3, x mol NaCl, y-x mol Na2CO3 dư
-Khi x = y: chỉ xẩy ra pư(1)
Dd C gồm x mol NaCl, x mol NaHCO3
-Khi x>y: pư (1) kết thúc và xẩy ra cả pư(2)
nNaCl = nNaHCO3 = nNa2CO3 = y mol
nHCl(1) = y mol, nHCl(2) = x-y mol
. Nếu x-y = y « x=2y pư(2) vừa đủ
ddC gồm: y mol NaCl(1) + y mol NaCl(2) = 2y mol
.Nếu x-y>y « x>2y dư HCl
Dd C gồm: y mol NaCl(1) + y mol NaCl(2) = 2y mol
HCl dư x-y-y = x-2y mol
. Nếu x-y< 2y « x<2y thì NaHCO3 dư
Dd C gồm: y mol NaCl(1) + x-y mol NaCl(2) = x mol
NaHCO3 dư: y-(x-y) = 2y-x mol
Bài 2: Cho 12 g MgO tác dụng vừa đủ với m g dd HCl 10,95 % thu được dd A
a, Tính khối lượng muối sinh ra và giá trị của m?
b, Tính C% ddA
c, Cho 400 ml dd NaOH vào dd A. Sau khi pư kết thúc, lọc bỏ kết tủa thu được dd nước lọc B. Nồng độ mol của các chất tan trong dd B bằng nhau. Tính CM dd NaOH đã dùng
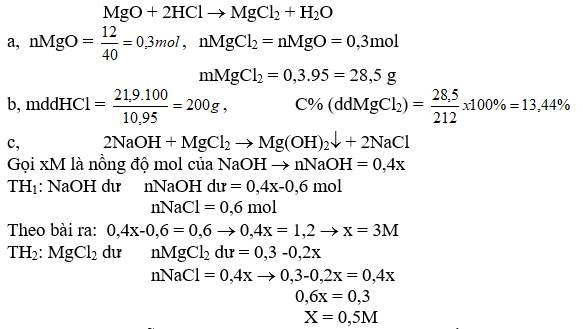
Bài 3: Cho 8,7 g hỗn hợp CuO và oxit kim loại(I) hòa tan hết vào 125 ml dd HCl 1,6M vừa đủ được ddA. Rót dd KOH vào dd A rồi lọc lấy kết tủa rửa sạch nung lên được chất rắn B. Thổi một luồng CO dư qua B nung nóng thu được 3,2 g chất rắn. Xác định KL(I)
Giải:
CuO + 2HCl ® CuCl2 + H2O (1)
M2O + 2HCl ® MCl + H2O (2)
CuCl2 + 2KOH ® Cu(OH)2 + 2KCl (3)
MCl + KOH ® MOH + KCl (4)
nHCl = 0,125 . 1,6 = 0,2 mol
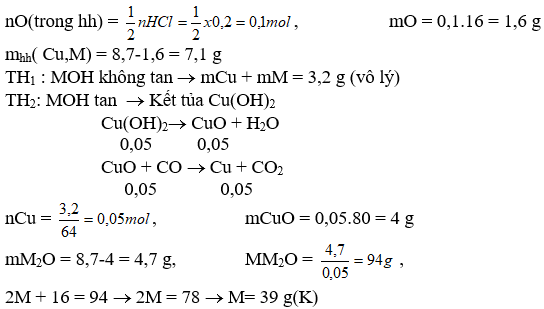
Bài 4: Cho 64 g Fe2O3 tác dụng vừa đủ với m g ddH2SO4 39,2% thu được ddA
a, Tính khối lượng muối sinh ra và giá trị của m
b, Tính C% của ddA
c, Cho 600 ml dd KOH vào dd A, sau khi phản ứng kết thúc, lọc bỏ kết tủa thu được dd nước lọc B. Nồng độ mol của các chất tan trong dd B bằng nhau
Tính CMdd KOH(bđ)
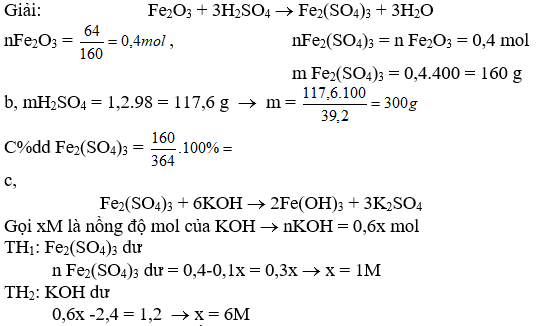
Bài 5: Cho 8 g CuO tan hết vào 200 ml dd H2SO4 được dd A. Cho dd A vào 300 ml dd Ba(OH)2 cùng nồng độ CM với dd H2SO4 ở trên được dd B và kết tủa C. Khối lượng kết tủa C là 56,4 g
a, Viết PTHH
b, Tính khối lượng mỗi chất trong C
c, Tính CMddB
Giải:
a, CuO + H2SO4 ® CuSO4 + H2O (1)
H2SO4 + Ba(OH)2 ® BaSO4 + 2H2O (2)
CuSO4 + Ba(OH)2 ® BaSO4 + Cu(OH)2 (3)
b, Gọi aM là nồng độ của dd H2SO4
n H2SO4 = 0,2a mol, nBa(OH)2 = 0,3a mol
TH1: H2SO4 hết ® dd A là CuSO4 « 0,2a = 0,1« a = 0,5M
mC = 233.0,1+ 9,8 = 33,1 g< 56,4 (loại)
TH2: H2SO4 dư
n H2SO4(1) = nCuO = 0,1 mol; nCuSO4 = 0,1 mol
n H2SO4(2) = 0,2a-0,1, nBa(OH)2(2) = 0,2a-0,1; nBa(OH)2(3) = 0,1a + 0,1
Theo pt(3): Ba(OH)2 dư
Bài ra: 233.(0,2a-0,1+0,1) + 9,8 = 56,4
46,6a = 46,6 ®a = 1M
c, dd B là dd Ba(OH)2 dư
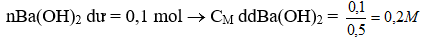
Bài 6: Hòa tan hoang toàn 16,4 g hỗn hợp X gồm MgO và MgCO3 trong dd H2SO4, sau phản ứng thu được dd A và 2,24 lit CO2(đktc)
a, Viết PTHH
b, Tính khối lượng mỗi chất trong 16,4 g hỗn hợp X
c, Cho 300 ml dd Ba(OH)2 1,5M vào dd A thu được 110,6 g kết tủa và 500 ml dd B.
Tính CM của chất trong dd B
Giải:
a, MgO + H2SO4 ® MgSO4 + H2O (1)
0,2 0,2
MgCO3 + H2SO4 ® MgSO4 + CO2 + H2O (2)
0,1 0,1
b, mMgCO3 = 0,1.84 = 8,4 g
mMgO = 16,4-8,4 = 8 g
c, nBa(OH)2 = 0,3.1,5 = 0,45 mol
TH1: H2SO4 hết, dd A là MgSO4
Ba(OH)2 + MgSO4 ® BaSO4 + Mg(OH)2 (3)
0,045 0,3 0,3 0,3
m kết tủa = 0,3.233 + 0,3.58 = 87,3 g <110,6 (loại)
TH2: H2SO4
Ba(OH)2 + H2SO4 ® BaSO4 + 2H2O
X x x
Ba(OH)2 + MgSO4 ® BaSO4 + Mg(OH)2
0,45-x 0,3
Gọi x là số mol của H2SO4 dư
. Nếu 0,45-x < 0,3« x> 0,15 thì MgSO4 dư
nMGSO4 dư = 0,3-( 0,45-x) = x-0,15
m kết tủa = 233.(x+ 0,45-x) + 58.(0,45-x) = 110,6
104,85 + 26,1 -58x = 110,6
x = 0,35
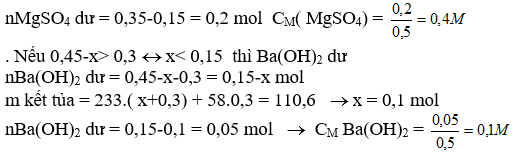
Giải: Khi cho từ từ HCl vào Na2CO3 xẩy ra các pư sau:
HCl + Na2CO3 ® NaHCO3 + NaCl (1)
x y y
HCl + NaHCO3 ® NaCl + CO2 + H2O (2)
x-y y
- Khi x<y : chỉ xẩy ra pư(1)
Dd C gồm x mol NaHCO3, x mol NaCl, y-x mol Na2CO3 dư
-Khi x = y: chỉ xẩy ra pư(1)
Dd C gồm x mol NaCl, x mol NaHCO3
-Khi x>y: pư (1) kết thúc và xẩy ra cả pư(2)
nNaCl = nNaHCO3 = nNa2CO3 = y mol
nHCl(1) = y mol, nHCl(2) = x-y mol
. Nếu x-y = y « x=2y pư(2) vừa đủ
ddC gồm: y mol NaCl(1) + y mol NaCl(2) = 2y mol
.Nếu x-y>y « x>2y dư HCl
Dd C gồm: y mol NaCl(1) + y mol NaCl(2) = 2y mol
HCl dư x-y-y = x-2y mol
. Nếu x-y< 2y « x<2y thì NaHCO3 dư
Dd C gồm: y mol NaCl(1) + x-y mol NaCl(2) = x mol
NaHCO3 dư: y-(x-y) = 2y-x mol
Bài 2: Cho 12 g MgO tác dụng vừa đủ với m g dd HCl 10,95 % thu được dd A
a, Tính khối lượng muối sinh ra và giá trị của m?
b, Tính C% ddA
c, Cho 400 ml dd NaOH vào dd A. Sau khi pư kết thúc, lọc bỏ kết tủa thu được dd nước lọc B. Nồng độ mol của các chất tan trong dd B bằng nhau. Tính CM dd NaOH đã dùng
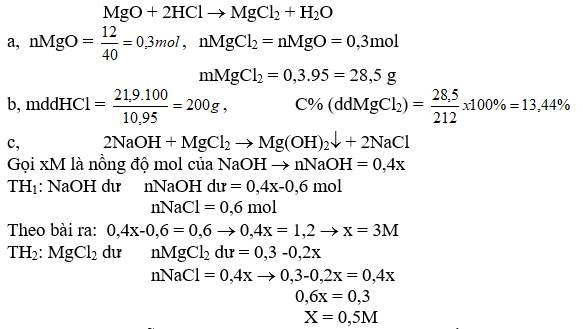
Bài 3: Cho 8,7 g hỗn hợp CuO và oxit kim loại(I) hòa tan hết vào 125 ml dd HCl 1,6M vừa đủ được ddA. Rót dd KOH vào dd A rồi lọc lấy kết tủa rửa sạch nung lên được chất rắn B. Thổi một luồng CO dư qua B nung nóng thu được 3,2 g chất rắn. Xác định KL(I)
Giải:
CuO + 2HCl ® CuCl2 + H2O (1)
M2O + 2HCl ® MCl + H2O (2)
CuCl2 + 2KOH ® Cu(OH)2 + 2KCl (3)
MCl + KOH ® MOH + KCl (4)
nHCl = 0,125 . 1,6 = 0,2 mol
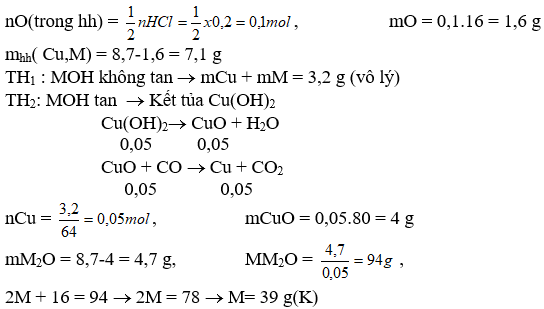
Bài 4: Cho 64 g Fe2O3 tác dụng vừa đủ với m g ddH2SO4 39,2% thu được ddA
a, Tính khối lượng muối sinh ra và giá trị của m
b, Tính C% của ddA
c, Cho 600 ml dd KOH vào dd A, sau khi phản ứng kết thúc, lọc bỏ kết tủa thu được dd nước lọc B. Nồng độ mol của các chất tan trong dd B bằng nhau
Tính CMdd KOH(bđ)
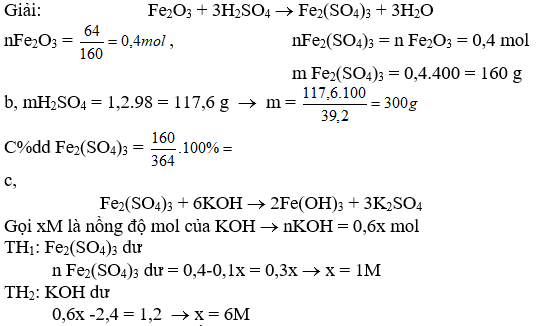
Bài 5: Cho 8 g CuO tan hết vào 200 ml dd H2SO4 được dd A. Cho dd A vào 300 ml dd Ba(OH)2 cùng nồng độ CM với dd H2SO4 ở trên được dd B và kết tủa C. Khối lượng kết tủa C là 56,4 g
a, Viết PTHH
b, Tính khối lượng mỗi chất trong C
c, Tính CMddB
Giải:
a, CuO + H2SO4 ® CuSO4 + H2O (1)
H2SO4 + Ba(OH)2 ® BaSO4 + 2H2O (2)
CuSO4 + Ba(OH)2 ® BaSO4 + Cu(OH)2 (3)
b, Gọi aM là nồng độ của dd H2SO4
n H2SO4 = 0,2a mol, nBa(OH)2 = 0,3a mol
TH1: H2SO4 hết ® dd A là CuSO4 « 0,2a = 0,1« a = 0,5M
mC = 233.0,1+ 9,8 = 33,1 g< 56,4 (loại)
TH2: H2SO4 dư
n H2SO4(1) = nCuO = 0,1 mol; nCuSO4 = 0,1 mol
n H2SO4(2) = 0,2a-0,1, nBa(OH)2(2) = 0,2a-0,1; nBa(OH)2(3) = 0,1a + 0,1
Theo pt(3): Ba(OH)2 dư
Bài ra: 233.(0,2a-0,1+0,1) + 9,8 = 56,4
46,6a = 46,6 ®a = 1M
c, dd B là dd Ba(OH)2 dư
Bài 6: Hòa tan hoang toàn 16,4 g hỗn hợp X gồm MgO và MgCO3 trong dd H2SO4, sau phản ứng thu được dd A và 2,24 lit CO2(đktc)
a, Viết PTHH
b, Tính khối lượng mỗi chất trong 16,4 g hỗn hợp X
c, Cho 300 ml dd Ba(OH)2 1,5M vào dd A thu được 110,6 g kết tủa và 500 ml dd B.
Tính CM của chất trong dd B
Giải:
a, MgO + H2SO4 ® MgSO4 + H2O (1)
0,2 0,2
MgCO3 + H2SO4 ® MgSO4 + CO2 + H2O (2)
0,1 0,1
b, mMgCO3 = 0,1.84 = 8,4 g
mMgO = 16,4-8,4 = 8 g
c, nBa(OH)2 = 0,3.1,5 = 0,45 mol
TH1: H2SO4 hết, dd A là MgSO4
Ba(OH)2 + MgSO4 ® BaSO4 + Mg(OH)2 (3)
0,045 0,3 0,3 0,3
m kết tủa = 0,3.233 + 0,3.58 = 87,3 g <110,6 (loại)
TH2: H2SO4
Ba(OH)2 + H2SO4 ® BaSO4 + 2H2O
X x x
Ba(OH)2 + MgSO4 ® BaSO4 + Mg(OH)2
0,45-x 0,3
Gọi x là số mol của H2SO4 dư
. Nếu 0,45-x < 0,3« x> 0,15 thì MgSO4 dư
nMGSO4 dư = 0,3-( 0,45-x) = x-0,15
m kết tủa = 233.(x+ 0,45-x) + 58.(0,45-x) = 110,6
104,85 + 26,1 -58x = 110,6
x = 0,35