PHƯƠNG TRÌNH HÓA HỌC
- Thứ bảy - 24/10/2020 10:31
- In ra
- Đóng cửa sổ này
Bài 1. Viết các phương trình hóa học xảy ra trong các thí nghiệm sau:

a) Nhỏ dung dịch Na2S vào dung dịch MgCl2
b) Sục khí H2S từ từ đến dư vào dung dịch FeCl3
c) Nhỏ dung dịch Na2CO3 đến dư vào dịch Fe2(SO4)3
d) Nhỏ dung dịch Na2S đến dư vào dung dịch AlCl3.
e) Nhỏ dung dịch NH3 đến dư vào dung dịch AlCl3.
f) Cho urê vào dung dịch Ba(OH)2 .
g) Hòa tan hỗn hợp gồm Cu và Fe2O3 vào dung dịch HCl dư.
h) Sục khí CO2 từ từ đến dư vào dung dịch hỗn hợp Ba(OH)2 và KOH
i) Nhỏ dung dịch NH3 dư vào các dung dịch CuCl2; ZnSO4; AgNO3
GIẢI
a) Na2S + MgCl2 + 2H2O ® Mg(OH)2 + 2NaCl + H2S
b) H2S + 2FeCl3 ® 2FeCl2 + S + 2HCl
c) Fe2(SO4)3 + 3Na2CO3 + 3H2O ® 2Fe(OH)3 + 3Na2SO4 + 3CO2
d) 3Na2S + 2AlCl3 + 6H2O ® 2Al(OH)3 + 6NaCl + 3H2S
e) 3NH3 + AlCl3 + 3H2O ® Al(OH)3 + 3NH4Cl
f) (NH2)2CO + 2H2O ® (NH4)2CO3
Ba(OH)2 + (NH4)2CO3 ® BaCO3 + 2NH3 + 2H2O
g) Fe2O3 + 6HCl ® 2FeCl3 + 3H2O
Cu + 2FeCl3 ® CuCl2 + 2FeCl2
h) Vì ban đầu CO2 thiếu nên thứ tự phản ứng xảy ra như sau:
Ba(OH)2 + CO2 ® BaCO3 + H2O
2KOH + CO2 ® K2CO3 + H2O
CO2 + K2CO3 + H2O ® 2KHCO3
CO2 + BaCO3 + H2O ® Ba(HCO3)2
i) CuCl2 + 2NH3 + 2H2O ® Cu(OH)2 + 2NH4Cl
Cu(OH)2 + 4NH3 ® Cu(NH3)4(OH)2
ZnSO4 + 2NH3 + 2H2O ® Zn(OH)2 + 2NH4Cl
Zn(OH)2 + 4NH3 ® Zn(NH3)4(OH)2
AgNO3 + 3NH3 + H2O ® Ag(NH3)2(OH) + NH4NO3
Bài 2. Viết PTHH xảy ra khi cho từ từ đến dư dung dịch:
a) NaOH vào dung dịch H3PO4
b) H3PO4 vào dung dịch NaOH.
c) AlCl3 vào dung dịch NaOH.
d) HCl vào dung dịch chứa Na2CO3 và NaHCO3
GIẢI
a) Vì ban đầu H3PO4 dư nên thứ tự phản ứng như sau:
NaOH + H3PO4 ® NaH2PO4 + H2O
NaOH + NaH2PO4 ® Na2HPO4 + H2O
NaOH + Na2HPO4 ® Na3PO4 + H2O
b) Vì ban đầu NaOH dư nên thứ tự phản ứng như sau:
H3PO4 + 3NaOH ® Na3PO4 + 3H2O
H3PO4 + 2Na3PO4 ® 3Na2HPO4
H3PO4 + Na2HPO4 ® 2NaH2PO4
c) Vì ban đầu NaOH dư nên Al(OH)3 vừa tạo ra ngay lập tức bị hòa tan.
AlCl3 + 3NaOH ® Al(OH)3 + 3NaCl
NaOH + Al(OH)3 ® NaAlO2 + 2H2O
AlCl3 + 3NaAlO2 + 6H2O ® 4Al(OH)3 + 3NaCl
d) Vì ban đầu HCl thiếu nên thứ tự phản ứng như sau:
Na2CO3 + HCl ® NaCl + NaHCO3
NaHCO3 + HCl ® NaCl + H2O + CO2
Bài 3. Cho dung dịch AlCl3 lần lượt tác dụng với các dung dịch sau:
a) NH3 b) Na2CO3 c) NaHCO3 d) NaAlO2
Viết các phương trình phản ứng xảy ra.
GIẢI
a) AlCl3 + 3NH3 + 3H2O ® Al(OH)3 + 3NH4Cl
+ 3NH4Cl
b) 2AlCl3 + 3Na2CO3 + 3H2O ® 2Al(OH)3 + 6NaCl + 3CO2
+ 6NaCl + 3CO2
c) AlCl3 + 3NaHCO3 ® Al(OH)3 + 3NaCl + 3CO2
+ 3NaCl + 3CO2
d) AlCl3 + 3NaAlO2 + 6H2O ® 4Al(OH)3 + 3NaCl
Bài 4. Viết phương trình phản ứng xảy ra khi cho từ từ đến dư:
a) Khí CO2 b) dung dịch NH4NO3. c) dung dịch NaHSO4
d) dung dịch HCl. e) dung dịch Al2(SO4)3
vào cốc chứa dung dịch KAlO2.
GIẢI
a) CO2 + 2KAlO2 + 3H2O ® K2CO3 + 2Al(OH)3
CO2 + K2CO3 + H2O ® 2KHCO3
.................................................................................................................................
CO2 + KAlO2 + 2H2O ® KHCO3 + Al(OH)3
b) KAlO2 + NH4NO3 + H2O ® Al(OH)3 + KNO3 + NH3
+ KNO3 + NH3
c) 2KAlO2 + 2NaHSO4 + 2H2O ® K2SO4 + Na2SO4 + 2Al(OH)3
2Al(OH)3 + 6NaHSO4 ® Al2(SO4)3 + 3Na2SO4 + 3H2O
d) KAlO2 + HCl + H2O ® 2KCl + Al(OH)3
Al(OH)3 + 3HCl ® AlCl3 + 3H2O
e) 6KAlO2 + Al2(SO4)3 + 12H2O ® 3K2SO4 + 8Al(OH)3
Chú ý: Muối KAlO2 là muối của axit rất yếu (HAlO2) với bazơ mạnh nên trong dung dịch bị thủy phân tạo môi trường kiềm: KAlO2 + HOH ® KOH + HAlO2
Đây là phản ứng thuận nghịch. Trong môi trường có sự hiện diện của một lượng nhỏ KOH.
Nếu có chất nào phản ứng được với KOH thì phản ứng chuyển dịch theo chiều thuận, là chiều tạo ra HAlO2. Chất này ngậm nước ( HAlO2.H2O) chính là Al(OH)3.
Bài 5. Viết phương trình phản ứng khi cho các dung dịch (mỗi dung dịch đều chứa 1 mol chất tan) tác dụng với nhau theo từng cặp sau:
a) BaCl2 và NaHSO4 b) Ba(HCO3)2 và KHSO4
c) Ca(H2PO4)2 và KOH d) Ca(OH)2 và NaHCO3
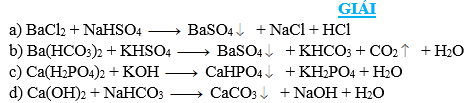
Bài 6. Viết các phương trình hóa học của các phản ứng xảy ra trong mỗi trường hợp sau:
a) Cho bột Cu vào dung dịch hỗn hợp NaNO3 và KHSO4.
b) Sục Cl2 vào dung dịch FeSO4 dư.
c) Sục Cl2 vào nước brom dư.
d) Nhỏ dung dịch Na2S2O3 vào dung dịch HCl.
e) Thêm nước vào clorua vôi rồi sục CO2 từ từ vào cho đến dư.
g) Cho Cu vào dung dịch hỗn hợp KNO3 và HCl.
h) Cho một mẩu Ba vào dung dịch NaHCO3.
GIẢI
a) 3Cu + 2NaNO3 + 8KHSO4 ® 3CuSO4 + Na2SO4 + 2NO + 4K2SO4 + 4H2O
b) 3Cl2 + 6FeSO4 ® 2Fe2(SO4)3 + 2FeCl3
c) 5Cl2 + Br2 + 6H2O ® 10HCl + 2HBrO3
d) H2SO4 + Na2S2O3 ® Na2SO4 + SO2 + S + H2O
e) CO2 + 2CaOCl2 + H2O ® CaCO3 + 2HClO + CaCl2
CO2 + CaCO3 + H2O ® Ca(HCO3)2
g) 3Cu + 2KNO3 + 8HCl ® 3CuCl2 + 2KCl + NO + 4H2O
e) Ba + 2H2O ® Ba(OH)2 + H2
Ba(OH)2 + 2NaHCO3 ® BaCO3 + Na2CO3 + 2H2O
Nếu Ba(OH)2 dư: Ba(OH)2 + Na2CO3 ® BaCO3 + 2NaOH
Bài 7. Viết các phương trình phản ứng xảy ra khi cho dung dịch NaHSO4 từ từ đến dư vào:
a) Fe b) Ba(NO3)2 c) Fe(OH)3 d) Na2CO3
e) NH4HSO3 g) FeS h) CuO i) NaAlO2
GIẢI
a) Fe + 2NaHSO4 ® FeSO4 + Na2SO4 + H2
b) Ba(NO3)2 + NaHSO4 ® BaSO4 + NaNO3 + HNO3
c) 2Fe(OH)3 + 6NaHSO4 ® Fe2(SO4)3 + 3Na2SO4 + 6H2O
d) Na2CO3 + NaHSO4 ® Na2SO4 + NaHCO3
NaHCO3 + NaHSO4 ® Na2SO4 + CO2 + H2O
e) NH4HSO3 + NaHSO4 ® Na2SO4+ (NH4)2SO4 + SO2 + H2O
g) FeS + NaHSO4 ® FeSO4 + Na2SO4 + H2S
h) CuO + 2NaHSO4 ® CuSO4 + Na2SO4 + H2O
i) NaAlO2 + NaHSO4 + H2O ® Na2SO4 + Al(OH)3
2Al(OH)3 + 6NaHSO4 ® Al2(SO4)3 + 3Na2SO4 + 6H2O
Chú ý: NaHSO4 hoặc KHSO4 có vai trò như H2SO4 loãng.
b) Sục khí H2S từ từ đến dư vào dung dịch FeCl3
c) Nhỏ dung dịch Na2CO3 đến dư vào dịch Fe2(SO4)3
d) Nhỏ dung dịch Na2S đến dư vào dung dịch AlCl3.
e) Nhỏ dung dịch NH3 đến dư vào dung dịch AlCl3.
f) Cho urê vào dung dịch Ba(OH)2 .
g) Hòa tan hỗn hợp gồm Cu và Fe2O3 vào dung dịch HCl dư.
h) Sục khí CO2 từ từ đến dư vào dung dịch hỗn hợp Ba(OH)2 và KOH
i) Nhỏ dung dịch NH3 dư vào các dung dịch CuCl2; ZnSO4; AgNO3
GIẢI
a) Na2S + MgCl2 + 2H2O ® Mg(OH)2 + 2NaCl + H2S
b) H2S + 2FeCl3 ® 2FeCl2 + S + 2HCl
c) Fe2(SO4)3 + 3Na2CO3 + 3H2O ® 2Fe(OH)3 + 3Na2SO4 + 3CO2
d) 3Na2S + 2AlCl3 + 6H2O ® 2Al(OH)3 + 6NaCl + 3H2S
e) 3NH3 + AlCl3 + 3H2O ® Al(OH)3 + 3NH4Cl
f) (NH2)2CO + 2H2O ® (NH4)2CO3
Ba(OH)2 + (NH4)2CO3 ® BaCO3 + 2NH3 + 2H2O
g) Fe2O3 + 6HCl ® 2FeCl3 + 3H2O
Cu + 2FeCl3 ® CuCl2 + 2FeCl2
h) Vì ban đầu CO2 thiếu nên thứ tự phản ứng xảy ra như sau:
Ba(OH)2 + CO2 ® BaCO3 + H2O
2KOH + CO2 ® K2CO3 + H2O
CO2 + K2CO3 + H2O ® 2KHCO3
CO2 + BaCO3 + H2O ® Ba(HCO3)2
i) CuCl2 + 2NH3 + 2H2O ® Cu(OH)2 + 2NH4Cl
Cu(OH)2 + 4NH3 ® Cu(NH3)4(OH)2
ZnSO4 + 2NH3 + 2H2O ® Zn(OH)2 + 2NH4Cl
Zn(OH)2 + 4NH3 ® Zn(NH3)4(OH)2
AgNO3 + 3NH3 + H2O ® Ag(NH3)2(OH) + NH4NO3
Bài 2. Viết PTHH xảy ra khi cho từ từ đến dư dung dịch:
a) NaOH vào dung dịch H3PO4
b) H3PO4 vào dung dịch NaOH.
c) AlCl3 vào dung dịch NaOH.
d) HCl vào dung dịch chứa Na2CO3 và NaHCO3
GIẢI
a) Vì ban đầu H3PO4 dư nên thứ tự phản ứng như sau:
NaOH + H3PO4 ® NaH2PO4 + H2O
NaOH + NaH2PO4 ® Na2HPO4 + H2O
NaOH + Na2HPO4 ® Na3PO4 + H2O
b) Vì ban đầu NaOH dư nên thứ tự phản ứng như sau:
H3PO4 + 3NaOH ® Na3PO4 + 3H2O
H3PO4 + 2Na3PO4 ® 3Na2HPO4
H3PO4 + Na2HPO4 ® 2NaH2PO4
c) Vì ban đầu NaOH dư nên Al(OH)3 vừa tạo ra ngay lập tức bị hòa tan.
AlCl3 + 3NaOH ® Al(OH)3 + 3NaCl
NaOH + Al(OH)3 ® NaAlO2 + 2H2O
AlCl3 + 3NaAlO2 + 6H2O ® 4Al(OH)3 + 3NaCl
d) Vì ban đầu HCl thiếu nên thứ tự phản ứng như sau:
Na2CO3 + HCl ® NaCl + NaHCO3
NaHCO3 + HCl ® NaCl + H2O + CO2
Bài 3. Cho dung dịch AlCl3 lần lượt tác dụng với các dung dịch sau:
a) NH3 b) Na2CO3 c) NaHCO3 d) NaAlO2
Viết các phương trình phản ứng xảy ra.
GIẢI
a) AlCl3 + 3NH3 + 3H2O ® Al(OH)3
 + 3NH4Cl
+ 3NH4Clb) 2AlCl3 + 3Na2CO3 + 3H2O ® 2Al(OH)3
 + 6NaCl + 3CO2
+ 6NaCl + 3CO2
c) AlCl3 + 3NaHCO3 ® Al(OH)3
 + 3NaCl + 3CO2
+ 3NaCl + 3CO2
d) AlCl3 + 3NaAlO2 + 6H2O ® 4Al(OH)3 + 3NaCl
Bài 4. Viết phương trình phản ứng xảy ra khi cho từ từ đến dư:
a) Khí CO2 b) dung dịch NH4NO3. c) dung dịch NaHSO4
d) dung dịch HCl. e) dung dịch Al2(SO4)3
vào cốc chứa dung dịch KAlO2.
GIẢI
a) CO2 + 2KAlO2 + 3H2O ® K2CO3 + 2Al(OH)3
CO2 + K2CO3 + H2O ® 2KHCO3
.................................................................................................................................
CO2 + KAlO2 + 2H2O ® KHCO3 + Al(OH)3
b) KAlO2 + NH4NO3 + H2O ® Al(OH)3
 + KNO3 + NH3
+ KNO3 + NH3
c) 2KAlO2 + 2NaHSO4 + 2H2O ® K2SO4 + Na2SO4 + 2Al(OH)3
2Al(OH)3 + 6NaHSO4 ® Al2(SO4)3 + 3Na2SO4 + 3H2O
d) KAlO2 + HCl + H2O ® 2KCl + Al(OH)3
Al(OH)3 + 3HCl ® AlCl3 + 3H2O
e) 6KAlO2 + Al2(SO4)3 + 12H2O ® 3K2SO4 + 8Al(OH)3
Chú ý: Muối KAlO2 là muối của axit rất yếu (HAlO2) với bazơ mạnh nên trong dung dịch bị thủy phân tạo môi trường kiềm: KAlO2 + HOH ® KOH + HAlO2
Đây là phản ứng thuận nghịch. Trong môi trường có sự hiện diện của một lượng nhỏ KOH.
Nếu có chất nào phản ứng được với KOH thì phản ứng chuyển dịch theo chiều thuận, là chiều tạo ra HAlO2. Chất này ngậm nước ( HAlO2.H2O) chính là Al(OH)3.
Bài 5. Viết phương trình phản ứng khi cho các dung dịch (mỗi dung dịch đều chứa 1 mol chất tan) tác dụng với nhau theo từng cặp sau:
a) BaCl2 và NaHSO4 b) Ba(HCO3)2 và KHSO4
c) Ca(H2PO4)2 và KOH d) Ca(OH)2 và NaHCO3
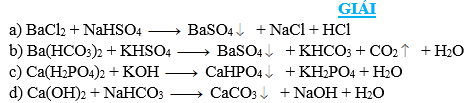
Bài 6. Viết các phương trình hóa học của các phản ứng xảy ra trong mỗi trường hợp sau:
a) Cho bột Cu vào dung dịch hỗn hợp NaNO3 và KHSO4.
b) Sục Cl2 vào dung dịch FeSO4 dư.
c) Sục Cl2 vào nước brom dư.
d) Nhỏ dung dịch Na2S2O3 vào dung dịch HCl.
e) Thêm nước vào clorua vôi rồi sục CO2 từ từ vào cho đến dư.
g) Cho Cu vào dung dịch hỗn hợp KNO3 và HCl.
h) Cho một mẩu Ba vào dung dịch NaHCO3.
GIẢI
a) 3Cu + 2NaNO3 + 8KHSO4 ® 3CuSO4 + Na2SO4 + 2NO + 4K2SO4 + 4H2O
b) 3Cl2 + 6FeSO4 ® 2Fe2(SO4)3 + 2FeCl3
c) 5Cl2 + Br2 + 6H2O ® 10HCl + 2HBrO3
d) H2SO4 + Na2S2O3 ® Na2SO4 + SO2 + S + H2O
e) CO2 + 2CaOCl2 + H2O ® CaCO3 + 2HClO + CaCl2
CO2 + CaCO3 + H2O ® Ca(HCO3)2
g) 3Cu + 2KNO3 + 8HCl ® 3CuCl2 + 2KCl + NO + 4H2O
e) Ba + 2H2O ® Ba(OH)2 + H2
Ba(OH)2 + 2NaHCO3 ® BaCO3 + Na2CO3 + 2H2O
Nếu Ba(OH)2 dư: Ba(OH)2 + Na2CO3 ® BaCO3 + 2NaOH
Bài 7. Viết các phương trình phản ứng xảy ra khi cho dung dịch NaHSO4 từ từ đến dư vào:
a) Fe b) Ba(NO3)2 c) Fe(OH)3 d) Na2CO3
e) NH4HSO3 g) FeS h) CuO i) NaAlO2
GIẢI
a) Fe + 2NaHSO4 ® FeSO4 + Na2SO4 + H2
b) Ba(NO3)2 + NaHSO4 ® BaSO4 + NaNO3 + HNO3
c) 2Fe(OH)3 + 6NaHSO4 ® Fe2(SO4)3 + 3Na2SO4 + 6H2O
d) Na2CO3 + NaHSO4 ® Na2SO4 + NaHCO3
NaHCO3 + NaHSO4 ® Na2SO4 + CO2 + H2O
e) NH4HSO3 + NaHSO4 ® Na2SO4+ (NH4)2SO4 + SO2 + H2O
g) FeS + NaHSO4 ® FeSO4 + Na2SO4 + H2S
h) CuO + 2NaHSO4 ® CuSO4 + Na2SO4 + H2O
i) NaAlO2 + NaHSO4 + H2O ® Na2SO4 + Al(OH)3
2Al(OH)3 + 6NaHSO4 ® Al2(SO4)3 + 3Na2SO4 + 6H2O
Chú ý: NaHSO4 hoặc KHSO4 có vai trò như H2SO4 loãng.