Bài tập về nguyên tử
- Thứ bảy - 17/10/2020 10:01
- In ra
- Đóng cửa sổ này
Dạng 1: TÍNH TOÁN VỀ NGUYÊN TỬ
●Phương pháp: ghi nhớ công thức
- Khối lượng tuyệt đối của nguyên tử: mnguyên tử =M .u với M là khôi lượng tương đối của nguyên tử so với u
●Phương pháp: ghi nhớ công thức
- Khối lượng tuyệt đối của nguyên tử: mnguyên tử =M .u với M là khôi lượng tương đối của nguyên tử so với u

- Khối lượng các hạt trong nguyên tử: u= 1,6605. 10-27 kg
me= 9,1 .10-31 kg = 0,0055u
mp= mn =1,66.10-27kg =u
- Tính khối lượng riêng: D=m/V với
●Bài tập mẫu:
Ví dụ 1: cho biết 1u = 1,6605. 10-27kg, nguyên tử khối của oxi bằng 15,999. hãy tính khối lượng tuyệt đối của oxi ra kg.
Giải:
Ta có: mo= M . u= 15,999.1,6605,10-27 = 26,566.10-27 kg
Ví dụ 2: trong 1,5kg đồng có bao nhiêu gam e ? cho biết 1 mol nguyên tử đồng có khối lượng bằng 63,546 gam, một nguyên tử đồng có 29 electron.
Giải:
Trong 63,546 gam có 6,02.1023 nguyên tử đồng
Ma: cứ 1 nguyên tử đồng có 29 e
Vậy: trong 63,546 gam đồng có 29.6,02.1023 e
Suy ra: trong 1500 gam đồng có 4121.1023 e
Suy ra khối lượng e trong 1500 gam đồng là:
4121.1023 .9,1.10-31=3,75.10-4 kg

●Bài tập mẫu:
Ví dụ 1 : Tổng số proton,electron,notron của nguyên tố X là 21.hãy xác định cấu tạo nguyên tử, gọi tên và viết kí hiệu nguyên tố X.
Giải:
Ta có: p+n+e=21 <=> 2p+n=21
<=>n=21-2p (1)
Mà: p≤n≤1,52p (2)
Từ (1)(2) =>3p≤21≤3,52p.
Thay : p=5 loại
p=6 loại vì A=9+6=15,không có trong BHTTH
p=8 loại
p=7 nhận vì A=7+7=14 là Nito
vậy p=n=e=7 và 714 N.
Ví dụ 2: Một kim loại M có số khối là 54,tổng số hạt p n e trong ion M2+ là 78.kí hiệu nguyên tử M là
A. 24Cr B. 25Mn C . 26Fe D. 27Co
Giải:
Theo giả thiết : p +n = 54
2p +n =80
Dạng 4:VIẾT CẤU HÌNH ELECTRON NGUYÊN TỬ, LIÊN QUAN GIỮA CẤU HÌNH ELECTRON
VÀ TÍNH CHẤT NGUYÊN
●Phương pháp :
- Xác định số e nguyên tử
-Các e được phân bố theo thứ tự tăng dần các mức năng lượng AO, theo các nguyên lý v quy tắc phn bố electron trong nguyn tử .
-Viết cấu hình electron theo thứ tự cc phn lớp trong một lớp v theo thứ tự của cc e.
●Bi tập mẫu :
Ví dụ 1 : Viết cấu hình electron của cc ion Cu2+, N3-, Fe3+,Cl- , Al3+. Biết số thứ tự của các nguyên tố lần lượt là Cu(Z=21), N(Z=29), Fe(Z=26), Cl(Z=17), Al(Z=13).
Giải:
Cu2+ : 1s2 2s2 2p6 3s2 3 p6 3d9
N3- : 1s2 2s2 2p6
Fe3+ : 1s2 2s2 2p6 3s2 3 p6 3d5
Cl- : 1s2 2s2 2p6 3s2 3 p6
Al3+: 1s2 2s2 2p6
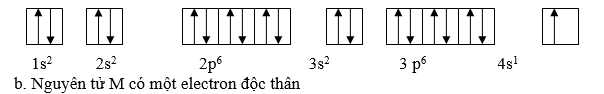
Ví dụ 2:Cho nguyên tử M khi mất đi một electron ta được cation M+ cĩ cấu hình electron ở lớp ngồi cng l 3p6 .
a. Viết cấu hình electron v sự phn bố electron theo Obitan của nguyn tử M
b. Cho biết số electron độc thân của nguyn tử M
Giải
Vì cation M+ được tạo thành do nguyên tử M mất một electron ở lớp vỏ ngoài cùng do đó nguyên tử M có một electron so với số electron 3p6 của M+
me= 9,1 .10-31 kg = 0,0055u
mp= mn =1,66.10-27kg =u
- Tính khối lượng riêng: D=m/V với
●Bài tập mẫu:
Ví dụ 1: cho biết 1u = 1,6605. 10-27kg, nguyên tử khối của oxi bằng 15,999. hãy tính khối lượng tuyệt đối của oxi ra kg.
Giải:
Ta có: mo= M . u= 15,999.1,6605,10-27 = 26,566.10-27 kg
Ví dụ 2: trong 1,5kg đồng có bao nhiêu gam e ? cho biết 1 mol nguyên tử đồng có khối lượng bằng 63,546 gam, một nguyên tử đồng có 29 electron.
Giải:
Trong 63,546 gam có 6,02.1023 nguyên tử đồng
Ma: cứ 1 nguyên tử đồng có 29 e
Vậy: trong 63,546 gam đồng có 29.6,02.1023 e
Suy ra: trong 1500 gam đồng có 4121.1023 e
Suy ra khối lượng e trong 1500 gam đồng là:
4121.1023 .9,1.10-31=3,75.10-4 kg

●Bài tập mẫu:
Ví dụ 1 : Tổng số proton,electron,notron của nguyên tố X là 21.hãy xác định cấu tạo nguyên tử, gọi tên và viết kí hiệu nguyên tố X.
Giải:
Ta có: p+n+e=21 <=> 2p+n=21
<=>n=21-2p (1)
Mà: p≤n≤1,52p (2)
Từ (1)(2) =>3p≤21≤3,52p.
Thay : p=5 loại
p=6 loại vì A=9+6=15,không có trong BHTTH
p=8 loại
p=7 nhận vì A=7+7=14 là Nito
vậy p=n=e=7 và 714 N.
Ví dụ 2: Một kim loại M có số khối là 54,tổng số hạt p n e trong ion M2+ là 78.kí hiệu nguyên tử M là
A. 24Cr B. 25Mn C . 26Fe D. 27Co
Giải:
Theo giả thiết : p +n = 54
2p +n =80
- trong nguyên tử M: p=e=26
- n=28 .vậy nguyên tử M là : 26Fe
- Dạng 3 : TÍNH TỈ KHỐI, TÍNH PHẦN TRĂM ĐỒNG VỊ VÀ NGUYÊN TỬ KHỐI TRUNG BÌNH
- ●Phương pháp: nghi nhớ công thức tính
- Nguyên tử khối trung bình A:

- Ví dụ 1 : trong tự nhiên Hidro có hai đồng vị 1H 99,984% và 2H 0,016% và Clo có hai đồng vị 35Cl 75,77% và 37Cl 24,23%
- tính nguyên tử khối trung bình của mổi nguyên tố
- có thề tạo nên được bao nhiêu phân tử HCl từ hai nguyên tử của hai nguyên tố đó.
- Tính phân tử khối của mổi loại hợp chất nói trên
- Giải :

- Ví dụ 2 :
Cho 4,12g dung dịch muối NaX tác dụng vừa đủ với dung dịch AgNO3 thu được 7,52g kết tủa.Tính nguyn tử khối X v gọi tn .Nguyên tử X có hai đồng vị biết đồng vị thứ hai có số nơtron nhiều hơn đồng vị thứ nhất là 2 và phầm trăm của 2 đồng vị bằng nhau ,tính số khối của mỗi đồng vị ?
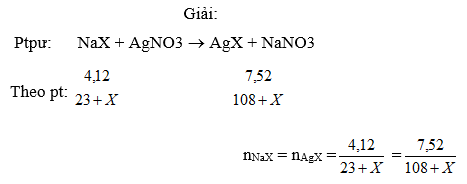

Dạng 4:VIẾT CẤU HÌNH ELECTRON NGUYÊN TỬ, LIÊN QUAN GIỮA CẤU HÌNH ELECTRON
VÀ TÍNH CHẤT NGUYÊN
●Phương pháp :
- Xác định số e nguyên tử
-Các e được phân bố theo thứ tự tăng dần các mức năng lượng AO, theo các nguyên lý v quy tắc phn bố electron trong nguyn tử .
-Viết cấu hình electron theo thứ tự cc phn lớp trong một lớp v theo thứ tự của cc e.
●Bi tập mẫu :
Ví dụ 1 : Viết cấu hình electron của cc ion Cu2+, N3-, Fe3+,Cl- , Al3+. Biết số thứ tự của các nguyên tố lần lượt là Cu(Z=21), N(Z=29), Fe(Z=26), Cl(Z=17), Al(Z=13).
Giải:
Cu2+ : 1s2 2s2 2p6 3s2 3 p6 3d9
N3- : 1s2 2s2 2p6
Fe3+ : 1s2 2s2 2p6 3s2 3 p6 3d5
Cl- : 1s2 2s2 2p6 3s2 3 p6
Al3+: 1s2 2s2 2p6
Ví dụ 2:Cho nguyên tử M khi mất đi một electron ta được cation M+ cĩ cấu hình electron ở lớp ngồi cng l 3p6 .
a. Viết cấu hình electron v sự phn bố electron theo Obitan của nguyn tử M
b. Cho biết số electron độc thân của nguyn tử M
Giải
Vì cation M+ được tạo thành do nguyên tử M mất một electron ở lớp vỏ ngoài cùng do đó nguyên tử M có một electron so với số electron 3p6 của M+
- Vậy cấu hình e v sự phn bố e vo AO :
